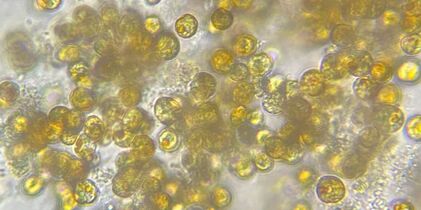
What are Zooxanthellae? And What is Coral Bleaching?
How Does REVIVE Work?
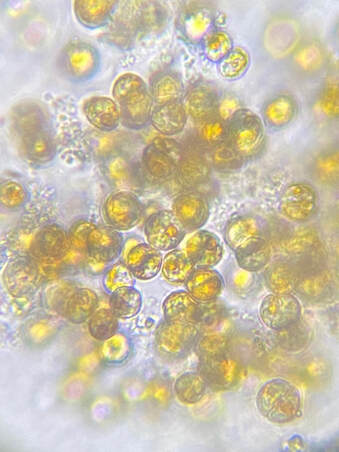
How to Use REVIVE REVIVE can be broadcast-fed or target-fed depending on the extent of the bleaching event. The necessary quantity and frequency of dosage is dependent on the severity and extent of the bleaching event as well as the water volume of the aquarium housing the affected corals. When using REVIVE as a broadcast-feed, we recommend a starting dosage of ¼ oz per 10 gallons added every day to the aquarium in a high flow area. Mechanical filtration should be disabled for approximately 2 hours after dosing REVIVE to allow the corals time to uptake the microalgae. Which Zooxanthellae were chosen for REVIVE?
A Closer Look at the Sub-Clades selected for REVIVE
REVIVE and Nourish In addition to zooxanthellae, REVIVE also contains the microalgae Rhodomonas sp. and Phaeodactylum tricornutum. Rhodomonas and Phaeodactylum are added to REVIVE as a supplemental live coral food intended to aid with the recovery of bleached corals through providing high quality nutrition. Aquarists often overlook the important role that live feeds play in the growth, coloration, and overall health of corals. While the zooxanthellae that reside within the tissues of the coral provide the coral with food, these zooxanthellae are only one component of nourishment for the coral. In order to sustain a vibrancy and optimal health, corals require regular planktonic feedings in addition to the food created by the zooxanthellae. Rhodomonas and Phaeodactylum have one of the highest microalgal polyunsaturated fatty acid concentrations making them a perfect addition to REVIVE for the purpose of nourishing your livestock back to health. Through dosing REVIVE, bleached corals can recapture their previously expelled zooxanthellae, and then these corals can feed on Rhodomonas and Phaeodactylum receiving the nourishment that they need to thrive. How Fast Does REVIVE Work?
References [1] Gomez, F., 2012. Problematic Biases in the Availability of Molecular Markers in Protists: The Example of the Dinoflagellates. Acta Protozoologica [2] Robinson, J. and M. Warner, 2006. Differential impacts of photoacclimation and thermal stress on the photobiology of four phylotypes of symbiotic dinoflagellates. Poster presentation, AGU meeting.
0 Comments
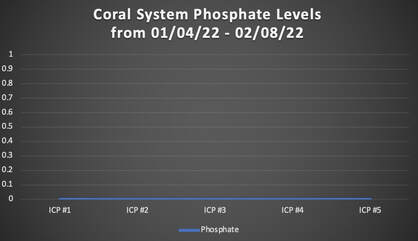
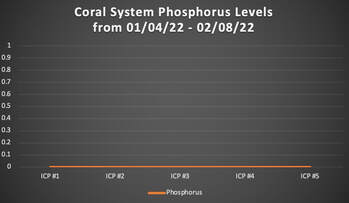
When we released REEF RECIPE™ (our premium dry coral food) in December of 2021, we made the claim that when REEF RECIPE is fed according to our instructions (see the REEF RECIPE label on the jar or the REEF RECIPE website page), no negative phosphate (PO4) affects should be observed. This claim that we made at REEF RECIPE's product release was based off of data collected from feedings here at our aquaculture facility coupled with data collected from beta-tester reef aquariums. Phosphate parameters were measured with a combination of both Hanna and Salifert test kits. Data collected both on- and off-site confirmed that when dosed according to our instructions, REEF RECIPE had no significant effect on phosphate levels in aquaria. In recent months, we have taken our REEF RECIPE phosphate experiment a step further. While the data previously collected via Hanna and Salifert test kits is undeniably valuable, we wanted to run a new feeding trial here out our aquaculture facility in which we dosed REEF RECIPE and sent off weekly water samples for Inductively Coupled Plasma - Optical Emission Spectrometry (ICP-OES) analysis to get the most accurate phosphate readings possible. Our methods and results are outlined below. Our REEF RECIPE Phosphate Experiment and Method ProtocolThe Experimental System Our experimental trial began on 01/04/2022 and was conducted through 02/08/2022. The system selected for the experiment was one of our coral systems here at our aquaculture facility. This particular coral system is approximately 850 gallons, and it houses a variety of corals (including soft corals, LPS, and SPS corals). Feeding Protocol The experimental coral system was fed once daily at a consistent dosage of 8.5 teaspoons, strictly following the instructions listed on the REEF RECIPE jar and the REEF RECIPE website page. For our experiment, we chose to broadcast feed (rather than target feed) the REEF RECIPE. Once the REEF RECIPE was hydrated in water from our coral system, we dumped the solution into a high-flow area of the coral system (i.e., in front of an Ecotech Vortech powerhead) to be dispersed to the corals. Data Collection Two water samples were collected from the experimental coral system between the hours of 10:00am EST and 3:00pm EST every Tuesday during the experimental trial. These water samples were mailed off same-day for ICP-OES analysis by Reef*Labs in Bradenton, FL. Specifically, we were interested in the ICP-OES readings for phosphate (PO4) and phosphorus. Results
Discussion - Nutrition without excess Nutrients and NuisanceOver the almost seven years that Aquaholic Aquaculture has been in business, we have tried feeding our corals a variety of different dry food brands that are on the market, but we had never been able to find a brand of dry food that performed up to our standards. Specifically we wanted to find a nutritionally balanced dry food, that contained a variety of both phytoplankton and zooplankton, that incited a feeding response from our corals, and that did NOT degrade water quality (specifically, did NOT significantly increase our phosphate levels). The most common issue that we found with other dry coral food brands on the market was that (even when feeding according to the manufacturer's instructions), our phosphate levels sky-rocketed, resulting in an abundance of unsightly green hair algae (GHA) and recurring red slime (cyanobacteria) blooms in our coral systems. Tired of battling nuisance algae and chasing low phosphate levels that were impossible to achieve whilst feeding these other brands of dry coral food, we decided to formulate our own dry coral food that would be just as nutritious without compromising water quality: REEF RECIPE. When formulating REEF RECIPE, we took both nutrition and water quality into account, and worked to create a food that would be beneficial and nutritional for all types of corals without causing water degradation. Since switching to feeding REEF RECIPE at our aquaculture facility, we have had no issue maintaining a phosphate level of 0 ppm, our corals have never looked better, and all of our green hair algae and red slime issues have completely disappeared. The results of our experiments confirm that aquarists can feed REEF RECIPE without compromising water quality or incurring the wrath of unwanted nuisance algae. For aquarists looking for a dry coral food that provides superior nutrition without water degradation from excess phosphates, REEF RECIPE is the coral food for your aquarium. Try REEF RECIPE today!The Pros and Cons of our Original 64 oz. Glass GrowlersAbout a year ago we added a new bulk-quantity packaging option for our REEFreshments: Live Phytoplankton and REEFreshments: REVIVE™ Live Zooxanthellae. The new packaging option was a 64 oz. glass growler. We chose these glass growlers, because we felt that they were the most appropriate choice of packaging for our #Aquaholic and #REEFreshments branding. What's more #Aquaholic than dosing live feeds from an amber glass growler, right?! And, once you finish feeding off your plankton, there's nothing stopping you from cleaning that growler and heading to your local brewery for a fill of your favorite brew! ;) For almost a year, we have had these glass growlers available through our website, retail and distribution partners, and at local frag swaps and trade shows. While overall our glass growlers have been a big hit with customers, we've received feedback that stores and customers would like the option to choose a more compact (and less fragile) packaging design than a glass growler. . . The Advantages of our NEW Dispenser Pump JugsOne of our favorite features of these new pump jugs is that they have measurement graduation markings clearly labeled on the side of the jug. These graduations make it easy to see just how much microalgae is left, and help simplify dosing. Like our glass growlers, these new jugs also feature a convenient handle for easy carrying and dosing. But! These new jugs ALSO come with the optional manual dispensing pump to make dosing a breeze! Just remove the cap, install the manual dispensing pump (included), and pump away! Don't Worry! Glass Growlers are Here to Stay!For all of our hardcore glass growlers fans, don't worry! The glass growlers are here to stay! We also love our glass growlers, so they're not going anywhere! That said, we also wanted to make sure that our REEFreshments packaging options accommodate customers looking for a more compact, less fragile container. For customers looking for more practical packaging, our new dispenser pump jugs are perfect! Try Our New Dispenser Pump Jugs Today!REEFreshments: Live Phytoplankton 64 oz. Dispenser Pump Jug - CLICK HERE
REEFreshments: REVIVE™ Live Zooxanthellae Dispenser Pump Jug - CLICK HERE
The Benefits of Dry Coral FoodsThere are two main merits of dry coral foods: (1) Convenience and (2) Cost-Effectiveness. (1) Convenience Our REEF RECIPE is easy to store (room temperature is sufficient), it has a one-year shelf life, and it is simple to feed. It doesn't get any easier than opening a jar of REEF RECIPE, hydrating a scoop of food (with tank water, RO water, or live phytoplankton), and feeding it to your reef aquarium. (2) Cost-Effectiveness Let's be honest, live feeds can be costly. While we recommend that all reef aquariums are fed live feeds (live plankton is the foundation to every marine ecosystem!), supplementing your live feed regimen with a dry coral food is a great way to feed your reef economically. Formulating REEF RECIPE™We have been growing live feeds for almost nine years, and producing them commercially for the aquarium industry for over six years now. Our experience in growing live feeds has been key in formulating our new dry coral food. Identifying which plankton are best suited for a dry coral food coupled with an understanding of the nutritional elements required for comprehensive reef nutrition, are the foundation for crafting a perfect dry coral food recipe. After formulating and testing a number of different dehydrated plankton-based coral food combinations, we refined our dry coral food ingredients for optimal nutrition creating: REEF RECIPE. REEF RECIPE™ - Refined NutritionREEF RECIPE consists of augmented marine plankton (both phytoplankton and zooplankton). Our recipe is a high-protein, carotenoid-rich formula with essential amino acids, minerals, vitamins, omega fatty acids (PUFAs and HUFAs), and immunostimulants. REEF RECIPE is designed to not just feed your reef, but nourish it. Our recipe has been refined to maximize coral growth, coloration, and health. What Makes REEF RECIPE™ Special(1) Variety of Micron Sizes & Suitable for ALL Corals REEF RECIPE contains a variety of micron sizes to ensure that there are appropriately sized food particles for every type of coral in your aquarium. From the smallest polyp corals (SPS) to the largest (LPS), REEF RECIPE has them (and every size in between) covered. It is also a great food for soft corals, corallimorphs, anemones, and other suspension-feeders. (2) High Protein Ranking in with a minimum crude protein percentage of 58%, REEF RECIPE has one of the highest protein counts of dry coral foods on the market. In comparison, some other dry coral foods only have about 30% crude protein. (3) PUFAs and HUFAs Polyunsaturated fatty acids (PUFAs) and highly unsaturated fatty acids (HUFAs) are essential to marine organism health and development. Most dry coral foods on the market contain polyunsaturated fatty acids (PUFAs), but not all of them contain highly unsaturated fatty acids (HUFAs). REEF RECIPE contains both PUFAs and HUFAs for optimal nutrition. (4) Carotenoid-Rich Carotenoids are fat-soluble pigments responsible for the vibrant colors that we see in corals and fish. Of the carotenoids commonly valued for marine pigmentation, Astaxanthin (a red-pigmented carotenoid) is especially prized for its color-enhancing properties. When formulating REEF RECIPE, we included marine plankton that are naturally high in carotenoids (some with natural Astaxanthin counts of ~1500ppm), and then we further augmented these plankton with CAROPHYLL® (a reputable carotenoid additive). CAROPHYLL® is unique in that it contains impressively high Astantaxanthin levels (~10%). In comparison, NatuRose® (an Astaxanthin supplement commonly used in other dry coral foods because of its substantially lower cost) is only about 1.5% Astaxanthin. At Aquaholic Aquaculture, we don't believe in sacrificing nutrition to save a little money. By choosing to augment our plankton with CAROPHYLL®, we offer almost 7x more Astaxanthin in our product than competitors who choose to supplement with NatuRose®. (5) Whole Plankton When planktonic food is dehydrated and pulverized, some of the nutritional value is lost in the process. In REEF RECIPE, you can clearly identify WHOLE copepods in the mix. We intentionally chose to keep these copepods intact so that they could retain their encapsulated nutrition, providing maximum nutrition for your reef. Plus, these large whole copepods are the perfect sized food for your larger polyp corals. (6) Minimal Water Degradation A common issue with feeding dry coral foods is water degradation. Many coral foods on the market contain high phosphates, which can be detrimental to a reef aquarium when dosed in large quantities. We took water quality into account when formulating REEF RECIPE, and can attest that our coral food only minimally affects phosphate levels. When dosed according to our instructions (see the REEF RECIPE label on the jar or the REEF RECIPE website page), no negative phosphate affects should be observed. Try REEF RECIPE™ Today!For the short and sweet version of why we love Phaeodactylum, click HERE for our announcement blog post: The New 5-Specie REEFreshments: Live Phytoplankton – Now with Phaeodactylum But! For those of you aquaholics who are like us and who love to geek-out about microalgae, you’re in the right place! Continue reading for our in-depth review of Phaeodactylum – The newest core specie of our now 5-specie REEFreshments: Live Phytoplankton blend! Our “Love Letter to Phaeodactylum tricornutum”The Specific Benefits that Phaeodactylum Brings to our Phytoplankton Blend and to Your Aquarium |
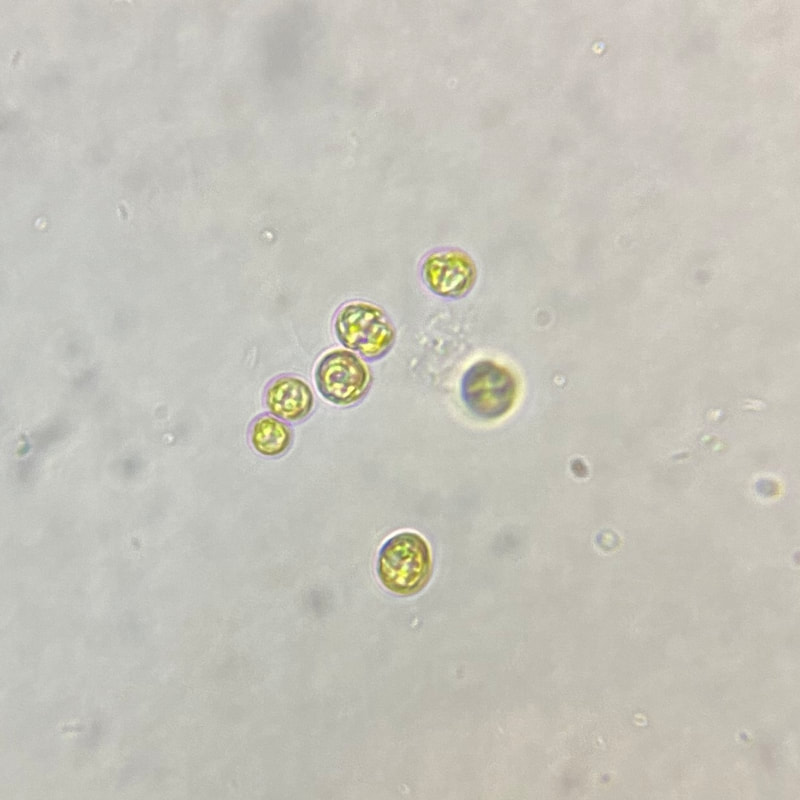
| Phaeodactylum tricornutum is an atypical species of phytoplankton. It is a marine, unicellular diatom, similar to Chaetoceros (also found in our REEFreshments phytoplankton blend), but it brings its own unique array of benefits, including: A robust nutritional profile, diversity in cell shape and size, a tolerance for reef-salinity, desirable phosphate uptake, and a non-reliance on the presence of silicates. | Microscopic image of P. tricornutum © Aquaholic Aquaculture |
(1) Highly Nutritious –
With Unparalleled Levels of Omega-3 Eicosapentaenoic Acid (EPA)
Phaeodactylum has long been used in mollusk and fish aquaculture because of its prized nutritional profile. Phaeodactylum boasts high-protein and high-carbohydrate ratios, and an exceptional Omega-3 fatty acid profile. Phaeodactylum provides impressive levels of essential highly unsaturated fatty acids (HUFAs) required to sustain health in marine organisms; specifically, it contains BOTH eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) (Hamilton et al., 2015).
Notably, Phaeodactylum’s EPA levels are exceptionally high, with EPA percentages reported as high as 51.8% (Cui et al., 2020). This EPA value far surpasses the EPA percentages for standard microalgae species cultured for use in the industry. With these remarkable EPA levels, Phaeodactylum significantly outperforms Nannochloropsis (which was formerly the ‘gold-standard’ of high-EPA phytoplankton) and earns its place as one of the most nutritious microalgae on the market.
How this Benefits Your Reef: Your reef organisms require complete and balanced nutrition to grow and thrive. Omega-3 HUFAs are of particular importance when it comes to marine organism health, with research demonstrating the critical role that EPA and DHA perform in sustaining marine life. Phaeodactylum contributes BOTH EPA and DHA to our blend (with exceptionally high levels of EPA), making our blend more balanced and nutritious than ever before!
Many phytoplankton products on the market are single-specie products that only provide either EPA or DHA, limiting the nutritional value of the product. For example, a single-specie phytoplankton product of only Nannochloropsis would contain relatively high levels of EPA (maximum of ~30%), but 0% DHA (Hoff & Snell, 1987), meaning that your reef inhabitants would not receive any DHA from the phytoplankton product, negatively impacting their health and survival.
With our multi-specie REEFreshments: Live Phytoplankton blend, you never have to worry about not providing a balanced and nutritious live feed. And, while our former 4-specie live phyto blend provided excellent nutrition, the addition of Phaeodactylum to our blend further elevates the nutritional profile of our REEFreshments: Live Phytoplankton, making it significantly more nutritionally comprehensive and robust.
Notably, Phaeodactylum’s EPA levels are exceptionally high, with EPA percentages reported as high as 51.8% (Cui et al., 2020). This EPA value far surpasses the EPA percentages for standard microalgae species cultured for use in the industry. With these remarkable EPA levels, Phaeodactylum significantly outperforms Nannochloropsis (which was formerly the ‘gold-standard’ of high-EPA phytoplankton) and earns its place as one of the most nutritious microalgae on the market.
How this Benefits Your Reef: Your reef organisms require complete and balanced nutrition to grow and thrive. Omega-3 HUFAs are of particular importance when it comes to marine organism health, with research demonstrating the critical role that EPA and DHA perform in sustaining marine life. Phaeodactylum contributes BOTH EPA and DHA to our blend (with exceptionally high levels of EPA), making our blend more balanced and nutritious than ever before!
Many phytoplankton products on the market are single-specie products that only provide either EPA or DHA, limiting the nutritional value of the product. For example, a single-specie phytoplankton product of only Nannochloropsis would contain relatively high levels of EPA (maximum of ~30%), but 0% DHA (Hoff & Snell, 1987), meaning that your reef inhabitants would not receive any DHA from the phytoplankton product, negatively impacting their health and survival.
With our multi-specie REEFreshments: Live Phytoplankton blend, you never have to worry about not providing a balanced and nutritious live feed. And, while our former 4-specie live phyto blend provided excellent nutrition, the addition of Phaeodactylum to our blend further elevates the nutritional profile of our REEFreshments: Live Phytoplankton, making it significantly more nutritionally comprehensive and robust.
(2) Various Cell Shapes
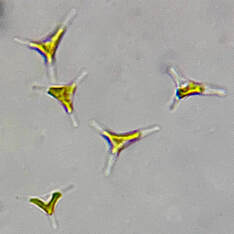
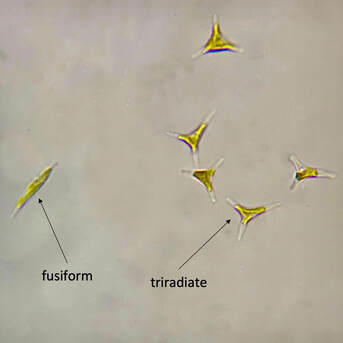
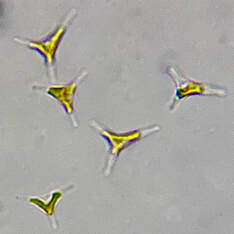
| One of the beloved oddities about Phaeodactylum is that the cells adapt to certain environmental conditions and change shape accordingly. Phaeodactylum can be found in oval, fusiform (peapod-shaped), triradiate (three-pointed-star), and the rare cruciform (cross) shape. It is theorized that Phaeodactylum changes shape to shift its buoyancy ability. Oval-shaped cells tend to clump together and sink, while fusiform, triradiate, and cruciform cells are more buoyant and better able stay in suspension. Research by Kuenzler and Ketchum (1962) and our own anecdotal experience culturing Phaeodactylum has demonstrated an increase in fusiform, triradiate, and cruciform (although rare) cells in lab cultures as these cell shapes are better favored by selection due to their buoyancy advantage. | Microscopic image of P. tricornutum © Aquaholic Aquaculture |
How this Benefits Your Reef: A phytoplankton better suited for staying in suspension means a food source for your reef that stays hovering in the water column ready for consumption by your hungry suspension-feeders (e.g., corals, clams, copepods, etc.). The better the suspension, the easier it is for suspension-feeders to consume the phytoplankton (Hentschel & Shimeta, 2008).
It also means that our new REEFreshments: Live Phytoplankton will better stay in suspension when bottled, which equates to a fresher, more viable product with an increased shelf life. It also means that you won’t need to shake your phyto bottles as frequently to keep the plankton from settling!
It also means that our new REEFreshments: Live Phytoplankton will better stay in suspension when bottled, which equates to a fresher, more viable product with an increased shelf life. It also means that you won’t need to shake your phyto bottles as frequently to keep the plankton from settling!
(3) Diverse Cell Sizes
As you may guess, because Phaeodactylum presents in various cell shapes, its cell sizes vary greatly. While most microalgae species cannot change their shape so their reported sizes are approximately the same, Phaeodactylum, with its variety of cell shapes, has a diverse range of cell sizes. On average, Phaeodactylum has a cell length of about 21μ and a cell width of about 4μ. However, Phaeodactylum cells have been reported as small as 3μ x 12μ, and as large as 5μ x 32μ.
How this Benefits Your Reef: Greater variety in cell shape and size improves prey-response. Not all suspension-feeders have the same dietary preferences, so the diversity of cell shapes and sizes increases the likelihood of consumption. The more variety in cell shape and size, the better the likelihood to appease all of your hungry reef inhabitants.
How this Benefits Your Reef: Greater variety in cell shape and size improves prey-response. Not all suspension-feeders have the same dietary preferences, so the diversity of cell shapes and sizes increases the likelihood of consumption. The more variety in cell shape and size, the better the likelihood to appease all of your hungry reef inhabitants.
(4) Suited for Reef Salinity
All of the phytoplankton species that we have selected for our REEFreshments: Live Phytoplankton blend thrive in reef-salinity parameters (~35ppt / ~1.025 sp gr), and Phaeodactylum is no exception, flourishing in reef-salinity environments (Hoff & Snell, 1987). In fact, research by Cui et al. (2020) demonstrated that Phaeodactylum’s growth rate was significantly greater at higher salinities (~35ppt).
How this Benefits Your Reef: One of the benefits of live phytoplankton over concentrated nonviable phytoplankton products is that live phytoplankton will live and grow in your aquarium until it is consumed by suspension-feeders (or removed by mechanical filtration or ultraviolet radiation). Live phyto has the advantage of continuously feeding your reef while also performing key nutrient export through the process of photosynthesis. While the live phytoplankton lives and reproduces in your aquarium, it consumes nitrates (NO3), phosphates (PO4), and carbon dioxide (CO2), while simultaneously producing oxygen (O2) for the aquarium. This nutrient export process reduces the phosphate and nitrate levels in the aquarium, which in turn helps to outcompete nuisance algae (e.g., hair algae, Bryopsis, red slime, etc.). Furthermore, by converting carbon dioxide to oxygen, the live phytoplankton helps maintain stable pH values by oxygenating the aquarium.
In order to reap all of the above benefits of live phyto, the phytoplankton species must be able to thrive and grow in reef aquarium environments, and, just like our original four core phyto species, Phaeodactylum thrives in these conditions.
How this Benefits Your Reef: One of the benefits of live phytoplankton over concentrated nonviable phytoplankton products is that live phytoplankton will live and grow in your aquarium until it is consumed by suspension-feeders (or removed by mechanical filtration or ultraviolet radiation). Live phyto has the advantage of continuously feeding your reef while also performing key nutrient export through the process of photosynthesis. While the live phytoplankton lives and reproduces in your aquarium, it consumes nitrates (NO3), phosphates (PO4), and carbon dioxide (CO2), while simultaneously producing oxygen (O2) for the aquarium. This nutrient export process reduces the phosphate and nitrate levels in the aquarium, which in turn helps to outcompete nuisance algae (e.g., hair algae, Bryopsis, red slime, etc.). Furthermore, by converting carbon dioxide to oxygen, the live phytoplankton helps maintain stable pH values by oxygenating the aquarium.
In order to reap all of the above benefits of live phyto, the phytoplankton species must be able to thrive and grow in reef aquarium environments, and, just like our original four core phyto species, Phaeodactylum thrives in these conditions.
(5) Great at Reducing Phosphate Levels
While all live phytoplankton help reduce phosphate levels in the aquarium, research has demonstrated Phaeodactylum to be exceptionally adept at phosphate consumption. In an experiment by Kuenzler and Ketchum (1962), the research team added phosphorous-deficient cultures of Phaeodactylum to phosphorous-rich controlled environments to analyze Phaeodactylum’s phosphate depletion rates. Their research found that upon introduction of Phaeodactylum to a phosphorous-rich environment, the microalgae immediately began phosphate uptake, resulting in a significant phosphate reduction to below measurable levels within only a few hours.
How this Benefits Your Reef: High phosphate levels are a problem that plagues many aquariums. Overfeeding, heavy bioloads, and the feeding of high-phosphate foods (among other factors) elevate phosphate levels in the aquarium which can become increasingly problematic. If phosphate levels are not kept in check, nuisance algae will begin to grow in the aquarium. And, if phosphate levels are not reduced in a timely manner, this nuisance algae can quickly overtake the aquarium, covering rocks, sand, glass, and even smothering corals and other marine sessile invertebrates. Even more worrisome, if phosphate levels climb too high, corals will begin to bleach and eventually die.
Phaeodactylum’s exceptional ability to consume phosphates means that dosing Phaeodactylum as part of our new 5-specie phytoplankton blend will help keep your phosphate levels in check, thereby reducing nuisance algae in the aquarium and fostering the continued health of your corals and other marine life.
How this Benefits Your Reef: High phosphate levels are a problem that plagues many aquariums. Overfeeding, heavy bioloads, and the feeding of high-phosphate foods (among other factors) elevate phosphate levels in the aquarium which can become increasingly problematic. If phosphate levels are not kept in check, nuisance algae will begin to grow in the aquarium. And, if phosphate levels are not reduced in a timely manner, this nuisance algae can quickly overtake the aquarium, covering rocks, sand, glass, and even smothering corals and other marine sessile invertebrates. Even more worrisome, if phosphate levels climb too high, corals will begin to bleach and eventually die.
Phaeodactylum’s exceptional ability to consume phosphates means that dosing Phaeodactylum as part of our new 5-specie phytoplankton blend will help keep your phosphate levels in check, thereby reducing nuisance algae in the aquarium and fostering the continued health of your corals and other marine life.
(6) No Silicate; No Problem
Most diatoms require the presence of silicate to build their cell walls, but not Phaeodactylum! In comparison with other diatoms, Phaeodactylum is weakly silicified (Kuenzler & Ketchum, 1962). In fact, research has demonstrated no observable difference in the growth rate of Phaeodactylum cells regardless of the presence of a silicate supplement (Cui et al., 2020).
How this Benefits Your Reef: The majority of established reef aquariums do not have high silicate levels, which means that most diatoms cannot grow and thrive in these long-established aquariums. However, since Phaeodactylum does not require the presence of silicate, it will thrive in established reef tanks!
How this Benefits Your Reef: The majority of established reef aquariums do not have high silicate levels, which means that most diatoms cannot grow and thrive in these long-established aquariums. However, since Phaeodactylum does not require the presence of silicate, it will thrive in established reef tanks!
REEFreshments: Live Phytoplankton –
A Refined Recipe for a Better Blend
We are beyond excited to be introducing Phaeodactylum to our REEFreshments: Live Phytoplankton blend. The array of benefits of Phaeodactylum make it an excellent candidate to join our flagship REEFreshments core phytoplankton species. Refining our proprietary REEFreshments: Live Phytoplankton blend into a new 5-specie blend through the addition of Phaeodactylum, has resulted in a superior phytoplankton product that we are proud to offer to the aquarium industry.
Since the introduction of our REEFreshments: Live Phytoplankton to the market in April of 2015, we have built a reputation for providing aquarists with a top-quality, live, viable, all-natural, nutritious, dense, multi-specie blend of phytoplankton. The addition of Phaeodactylum to our REEFreshments: Live Phytoplankton blend refines our recipe to ensure that we continue to provide a superior phytoplankton product. Rest assured that you can trust and rely on REEFreshments for your reefing endeavors, as we will always commit to producing live feeds of the highest standards of quality and excellence.
We know that you and your reef aquarium will love Phaeodactylum and our new 5-specie blend of REEFreshments: Live Phytoplankton as much as we do. As always, it is our pleasure to feed your reef. Cheers!
Click HERE to try our new 5-specie phytoplankton blend!
Since the introduction of our REEFreshments: Live Phytoplankton to the market in April of 2015, we have built a reputation for providing aquarists with a top-quality, live, viable, all-natural, nutritious, dense, multi-specie blend of phytoplankton. The addition of Phaeodactylum to our REEFreshments: Live Phytoplankton blend refines our recipe to ensure that we continue to provide a superior phytoplankton product. Rest assured that you can trust and rely on REEFreshments for your reefing endeavors, as we will always commit to producing live feeds of the highest standards of quality and excellence.
We know that you and your reef aquarium will love Phaeodactylum and our new 5-specie blend of REEFreshments: Live Phytoplankton as much as we do. As always, it is our pleasure to feed your reef. Cheers!
Click HERE to try our new 5-specie phytoplankton blend!
References
[1] Cui, Y., Thomas‐Hall, S. R., Chua, E. T., & Schenk, P. M. (2020). Development of high‐level omega‐3 eicosapentaenoic acid (EPA) production from Phaeodactylum Tricornutum. Journal of Phycology, 57(1), 258–268.
[2] Hamilton, M. L., Warwick, J., Terry, A., Allen, M. J., Napier, J. A., & Sayanova, O. (2015). Towards the industrial production of omega-3 long chain polyunsaturated fatty acids from a genetically modified diatom Phaeodactylum tricornutum. PLOS ONE, 10(12).
[3] Hentschel, B. T., & Shimeta, J. (2008). Suspension Feeders. Encyclopedia of Ecology.
[4] Hoff, F. H., & Snell, T. W. (1987). Plankton culture manual. Florida Aqua Farms Inc.
[5] Kuenzler, E. J., & Ketchum, B. H. (1962). Rate of phosphorus uptake by Phaeodactylum tricornutum. The Biological Bulletin, 123(1), 134–145.
[2] Hamilton, M. L., Warwick, J., Terry, A., Allen, M. J., Napier, J. A., & Sayanova, O. (2015). Towards the industrial production of omega-3 long chain polyunsaturated fatty acids from a genetically modified diatom Phaeodactylum tricornutum. PLOS ONE, 10(12).
[3] Hentschel, B. T., & Shimeta, J. (2008). Suspension Feeders. Encyclopedia of Ecology.
[4] Hoff, F. H., & Snell, T. W. (1987). Plankton culture manual. Florida Aqua Farms Inc.
[5] Kuenzler, E. J., & Ketchum, B. H. (1962). Rate of phosphorus uptake by Phaeodactylum tricornutum. The Biological Bulletin, 123(1), 134–145.
For over six years we have provided our customers with a top-quality live phytoplankton feed. REEFreshments: Live Phytoplankton has become a staple for aquarists all over the United States. The positive feedback and success stories that we receive from aquarists dosing our phytoplankton is the best reward we could ask for, and it fuels our passion to continue producing live feeds at the highest standards of quality and excellence.
While our REEFreshments: Live Phytoplankton blend is tried and true, we are constantly looking for ways to improve our products and make sure that we continue to provide aquarists with superior live feed blends. With that as our mission, we are excited to announce a refined REEFreshments: Live Phytoplankton recipe that now contains an extra specie of phytoplankton, making our phyto now a 5-specie blend!
While our REEFreshments: Live Phytoplankton blend is tried and true, we are constantly looking for ways to improve our products and make sure that we continue to provide aquarists with superior live feed blends. With that as our mission, we are excited to announce a refined REEFreshments: Live Phytoplankton recipe that now contains an extra specie of phytoplankton, making our phyto now a 5-specie blend!
| Introducing our new specie addition: Phaeodactylum tricornutum! That’s right! The best live phyto on the market just got better! Our REEFreshments: Live Phytoplankton blend will continue to include our four core microalgae strains (Chaetoceros, Isochrysis, Nannochloropsis, and Tetraselmis), but will now also include the marine, unicellular, motile diatom Phaeodactylum! With the addition of Phaeodactylum, our now 5-specie blend will provide increased cell diversity, augmented nutrition, and an enhanced array of other benefits for your aquarium inhabitants! | Microscopic image of P. tricornutum © Aquaholic Aquaculture |
Why We Decided to Refine our Phyto Recipe
Let me start by saying, we did not take revising our phytoplankton blend lightly. The decision to add Phaeodactylum to our REEFreshments blend was only made after much research on this particular species of microalgae. This research included: An extensive review of the literature on Phaeodactylum, experimentation with culturing this microalgae in our Phyto Lab at Aquaholic Aquaculture, and regular dosing of Phaeodactylum in our own coral systems at Aquaholic as well as in beta-tester reef aquariums over the course of the last year.
Our research and experiments have concluded the following: Phaeodactylum is a unique, nutritious, and hardy microalgae that deserves a place alongside our core microalgae strains that comprise our REEFreshments phytoplankton blend. The addition of Phaeodactylum to our REEFreshments: Live Phytoplankton only makes our blend BETTER! We are in LOVE with Phaeodactylum and are beyond excited to be introducing it as our new 5th core phyto strain!
Our research and experiments have concluded the following: Phaeodactylum is a unique, nutritious, and hardy microalgae that deserves a place alongside our core microalgae strains that comprise our REEFreshments phytoplankton blend. The addition of Phaeodactylum to our REEFreshments: Live Phytoplankton only makes our blend BETTER! We are in LOVE with Phaeodactylum and are beyond excited to be introducing it as our new 5th core phyto strain!
The Short and Sweet of Why We LOVE Phaeodactylum
1. Highly Nutritious – Phaeodactylum boasts an excellent nutritional profile. It features high-protein and high-carbohydrate ratios, and an exceptional Omega-3 fatty acid profile. Phaeodactylum provides impressive levels of essential highly unsaturated fatty acids (HUFAs) required to sustain health in marine organisms; specifically, it contains BOTH eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Notably, Phaeodactylum’s EPA value (reported as high as 51.8%) far surpasses the EPA percentages for standard microalgae species cultured for use in the industry.
2. Various Cell Shapes and Sizes – Phaeodactylum is unique from most other microalgae in that it can change its cell shape and size to adjust its cell buoyancy ratios and adapt to environmental conditions. Because of this, Phaeodactylum incites an increased prey-response from suspension-feeders (e.g., corals, clams, copepods, etc.). Not all suspension-feeders have the same dietary preferences, so the diversity of cell shapes and sizes increases the likelihood of consumption.
3. Adapted Cell Buoyancy and Increased Suspension – Phaeodactylum’s ability to change its shape to increase its cell buoyancy lends it to have superior suspension properties in comparison to other microalgae. In your aquarium, this means that the phytoplankton will stay better suspended in the water column enabling easier consumption by suspension-feeders. In refrigerated storage, this means better suspension in packaging which equates to: A more nutritious and viable feed, extended shelf life, and the reduced need to shake bottles as frequently in order to keep the phyto in suspension.
4. Thrives in Reef Aquarium Conditions – Like our other REEFreshments phytoplankton core species, Phaeodactylum readily grows and thrives at reef-salinity (~35ppt / ~1.025 sp gr). Furthermore, unlike other diatoms, Phaeodactylum does not require the presence of silicate to survive and grow, which means that it will excel even in long-established, silicate-depleted reef aquariums.
5. Excellent Phosphate Uptake – Research has demonstrated Phaeodactylum’s excellence in phosphorous consumption. This means that dosing Phaeodactylum in your aquarium will help reduce phosphate levels, and, in turn, help outcompete nuisance algae.
2. Various Cell Shapes and Sizes – Phaeodactylum is unique from most other microalgae in that it can change its cell shape and size to adjust its cell buoyancy ratios and adapt to environmental conditions. Because of this, Phaeodactylum incites an increased prey-response from suspension-feeders (e.g., corals, clams, copepods, etc.). Not all suspension-feeders have the same dietary preferences, so the diversity of cell shapes and sizes increases the likelihood of consumption.
3. Adapted Cell Buoyancy and Increased Suspension – Phaeodactylum’s ability to change its shape to increase its cell buoyancy lends it to have superior suspension properties in comparison to other microalgae. In your aquarium, this means that the phytoplankton will stay better suspended in the water column enabling easier consumption by suspension-feeders. In refrigerated storage, this means better suspension in packaging which equates to: A more nutritious and viable feed, extended shelf life, and the reduced need to shake bottles as frequently in order to keep the phyto in suspension.
4. Thrives in Reef Aquarium Conditions – Like our other REEFreshments phytoplankton core species, Phaeodactylum readily grows and thrives at reef-salinity (~35ppt / ~1.025 sp gr). Furthermore, unlike other diatoms, Phaeodactylum does not require the presence of silicate to survive and grow, which means that it will excel even in long-established, silicate-depleted reef aquariums.
5. Excellent Phosphate Uptake – Research has demonstrated Phaeodactylum’s excellence in phosphorous consumption. This means that dosing Phaeodactylum in your aquarium will help reduce phosphate levels, and, in turn, help outcompete nuisance algae.
Our extensive research has demonstrated Phaeodactylum to be the perfect new microalgae specie to add to our REEFreshments: Live Phytoplankton blend. The addition of Phaeodactylum makes our now 5-specie phyto blend more diverse, nutritious, and beneficial than ever before. We know that you and your reef aquarium will love Phaeodactylum as much as we do, and we are excited to hear about your experiences with our new enhanced 5-specie REEFreshments: Live Phytoplankton blend! Cheers!
Click HERE to try our new 5-specie phytoplankton blend!
Click HERE to try our new 5-specie phytoplankton blend!
P.S. We kept this announcement short and sweet, because we know that not everyone enjoys the science of microalgae as much as we do. But! For those of you aquaholics who are like us and who love to geek-out about microalgae, you can view our in-depth “Love Letter to Phaeodactylum tricornutum” HERE, in which we further delve into our research on this particular specie of microalgae and the specific benefits that it brings to reef aquariums.
References
[1] Cui, Y., Thomas‐Hall, S. R., Chua, E. T., & Schenk, P. M. (2020). Development of high‐level omega‐3 eicosapentaenoic acid (EPA) production from Phaeodactylum Tricornutum. Journal of Phycology, 57(1), 258–268.
[2] Hamilton, M. L., Warwick, J., Terry, A., Allen, M. J., Napier, J. A., & Sayanova, O. (2015). Towards the industrial production of omega-3 long chain polyunsaturated fatty acids from a genetically modified diatom Phaeodactylum tricornutum. PLOS ONE, 10(12).
[3] Hentschel, B. T., & Shimeta, J. (2008). Suspension Feeders. Encyclopedia of Ecology.
[4] Hoff, F. H., & Snell, T. W. (1987). Plankton culture manual. Florida Aqua Farms Inc.
[5] Kuenzler, E. J., & Ketchum, B. H. (1962). Rate of phosphorus uptake by Phaeodactylum tricornutum. The Biological Bulletin, 123(1), 134–145.
[2] Hamilton, M. L., Warwick, J., Terry, A., Allen, M. J., Napier, J. A., & Sayanova, O. (2015). Towards the industrial production of omega-3 long chain polyunsaturated fatty acids from a genetically modified diatom Phaeodactylum tricornutum. PLOS ONE, 10(12).
[3] Hentschel, B. T., & Shimeta, J. (2008). Suspension Feeders. Encyclopedia of Ecology.
[4] Hoff, F. H., & Snell, T. W. (1987). Plankton culture manual. Florida Aqua Farms Inc.
[5] Kuenzler, E. J., & Ketchum, B. H. (1962). Rate of phosphorus uptake by Phaeodactylum tricornutum. The Biological Bulletin, 123(1), 134–145.
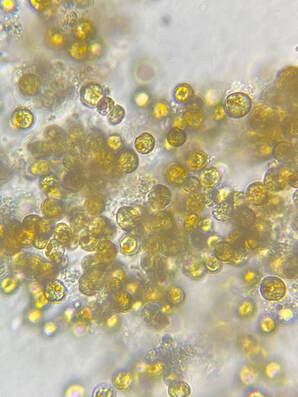
What is Phytoplankton?
| Microalgae, more commonly referred to as phytoplankton in the aquarium industry, are aquatic, microscopic, single-celled, photosynthetic organisms. Similar to plants (their land counterparts), phytoplankton have chlorophyll in their cells, and generate food through the process of photosynthesis. Phytoplankton are primary producers and form the foundation of virtually all marine food webs, supporting marine ecosystems and making other aquatic life possible. Both in the wild and in aquariums, Phytoplankton are an essential component to a balanced reef ecosystem. | Microscopic image of microalgae © Aquaholic Aquaculture |
Nutritional Value of Phytoplankton
Phytoplankton is an abundant source of macro- and micronutrients, including: Protein, carbohydrates, vitamins, lipids, and trace elements. Arguably one of the most important nutrients contained within phytoplankton cells are lipids, in particular Omega-3 fatty acids. Omega-3 fatty acids, often referred to as highly unsaturated fats (HUFA), are absolutely essential for the health and coloration of marine invertebrates and fish. Of these fatty acids, eicosapentaenoic acid or EPA (found generally in green-colored phytoplankton species) and docosahexaenoic acid or DHA (found generally in brown-colored phytoplankton species) are of critical importance to marine life, with DHA proving to be the more essential of the two.
When dosing phytoplankton in an aquarium, in order to obtain maximum nutrition, dosing multiple species with various nutritional profiles is important. A good dosing regimen for a reef aquarium is a MINIMUM of one species of a high-EPA (e.g., Nannochloropsis) and one specie of a high-DHA algae (e.g., Isochrysis) to ensure that the aquarium inhabitants receive proper nutrition.
When dosing phytoplankton in an aquarium, in order to obtain maximum nutrition, dosing multiple species with various nutritional profiles is important. A good dosing regimen for a reef aquarium is a MINIMUM of one species of a high-EPA (e.g., Nannochloropsis) and one specie of a high-DHA algae (e.g., Isochrysis) to ensure that the aquarium inhabitants receive proper nutrition.
What Consumes Phytoplankton?
In both the wild and in aquariums, phytoplankton is a perfect food for: Corals, clams, feather dusters, scallops, tunicates and sponges, copepods, tube-worms, sea squirts, and other suspension-feeding invertebrates. In aquariums, regular dosing of phytoplankton increases the health, coloration, growth, and polyp extension of corals, increases the development and vibrancy of your marine life, and boosts zooplankton populations. You can view our Live Feed Guide for a more thorough breakdown of the marine life that benefit from phytoplankton dosing.
Maintaining Water Quality
Phytoplankton play a major role in water quality and nutrient recycling through their consumption of nitrogenous wastes. Nitrogen and phosphorous are required by the phytoplankton to perform photosynthesis. In an aquarium environment, adding live phytoplankton will reduce your nitrate (NO3) and phosphate (PO4) levels as the living phytoplankton consume these nutrients for photosynthesis.
Not only will live phytoplankton help reduce NO3 and PO4 levels, but through photosynthesis the phytoplankton help balance pH levels by increasing dissolved oxygen levels in the aquarium while simultaneously reducing carbon dioxide levels.
How drastically can phytoplankton affect water quality? It turns out, quite a lot! In fact, because phytoplankton are so effective at nutrient uptake and because of their capacity for exponential growth, phytoplankton are sometimes utilized at municipal water treatment facilities to remove primary nutrients from sewage and wastewater. So, if phytoplankton can tackle sewage, just imagine the work that phytoplankton can do in your aquarium!
Not only will live phytoplankton help reduce NO3 and PO4 levels, but through photosynthesis the phytoplankton help balance pH levels by increasing dissolved oxygen levels in the aquarium while simultaneously reducing carbon dioxide levels.
How drastically can phytoplankton affect water quality? It turns out, quite a lot! In fact, because phytoplankton are so effective at nutrient uptake and because of their capacity for exponential growth, phytoplankton are sometimes utilized at municipal water treatment facilities to remove primary nutrients from sewage and wastewater. So, if phytoplankton can tackle sewage, just imagine the work that phytoplankton can do in your aquarium!
Outcompeting Nuisance Algae
A further benefit of dosing live phytoplankton in an aquarium is to help reduce or prevent nuisance algae growth (e.g., hair algae, bryopsis, cyano, etc.). Nuisance algae in aquariums require the same nutrients as live phytoplankton to grow and survive. If live phytoplankton is dosed regularly and consumes these nutrients, the nuisance algae will eventually wither away.
What to Look for in a Quality Phytoplankton Product
There are a lot of phytoplankton products on the aquarium market, and there is a lot of misinformation about what constitutes a quality phytoplankton product. If you are looking for a quality phytoplankton product that will give your reef the benefits of maximum nutrition, optimal water quality, and help reduce nuisance algae growth, then here's what you need to look for:
- MULTI-SPECIE BLEND
***For proper nutrition, you need to dose a MINIMUM of TWO species of phytoplankton.***
- EPA & DHA (GREENS & BROWNS)
***It is critical to dose BOTH high-EPA (typically "green") and high-DHA (typically "brown") species.***
- LIVE (in contrast to concentrates, pastes, or cryopreserved)
***In order to receive the benefits of nutrient consumption, the product must be LIVE.***
- NO PRESERVATIVES, GELS, BINDERS, OR OTHER ADDITIVES
***Avoid products with additives that could be potentially harmful to your reef.***
REEFreshments®: Live Phytoplankton
| Our REEFreshments®: Live Phytoplankton meets all of the above criteria for a quality phytoplankton product. Our phytoplankton is a MULTI-SPECIE BLEND, containing three high-EPA microalgae (Nannochloropsis, Phaeodactylum, and Tetraselmis) and two high-DHA microalgae (Isochrysis and Chaetoceros), it is LIVE, and it does NOT contains any preservatives, gels, binders, or other additives. For more specifics about our REEFreshments®: Live Phytoplankton and/or to purchase a bottle for your reef aquarium, CLICK HERE. |
References
[1] Delbeek, J. C., & Sprung, J. (2005). The Reef Aquarium: Science, Art, and Technology. Ricordea Pub.
[2] Hentschel, B. T., & Shimeta, J. (2008). Suspension Feeders. Encyclopedia of Ecology.
[3] Hoff, F. H., & Snell, T. W. (1987). Plankton culture manual. Florida Aqua Farms Inc.
[4] Levinton, J. S. (2009). Marine Biology: Function, biodiversity, ecology. Oxford University Press.
[5] Wittenrich, M. L. (2007). The complete illustrated breeder's guide to marine aquarium fishes. T.F.H.
[2] Hentschel, B. T., & Shimeta, J. (2008). Suspension Feeders. Encyclopedia of Ecology.
[3] Hoff, F. H., & Snell, T. W. (1987). Plankton culture manual. Florida Aqua Farms Inc.
[4] Levinton, J. S. (2009). Marine Biology: Function, biodiversity, ecology. Oxford University Press.
[5] Wittenrich, M. L. (2007). The complete illustrated breeder's guide to marine aquarium fishes. T.F.H.
Aquaholic Blog
Our blog is dedicated to educating aquarium hobbyists about all-things-aquaculture, as well as sharing our aquatic adventures at Aquaholic Aquaculture® with fellow #Aquaholics
Archives
February 2023
April 2022
December 2021
November 2021
August 2021








 RSS Feed
RSS Feed